National Molecular Pathology Board
Principal Investigator: Philipp Lange
Chair: Cynthia Hawkins
Vice-Chair: Jonathan Bush
Highly advanced assays are being rapidly developed and introduced, with significant potential to improve patient care. However, their visibility and accessibility vary widely across centres, and challenges remain in determining which assays are most appropriate for individual patients and how to access them. The National Molecular Pathology Board (MPB) aims to broaden and democratize access to advanced molecular testing for pediatric, adolescent, and young adult (AYA) cancer patients across Canada. By bringing together national experts in molecular pathology, the MPB provides a coordinated pathway for evaluating and recommending the most appropriate molecular assays for each referred case.
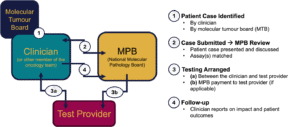
Through this initiative, patient cases with significant potential to benefit from advanced molecular analyses are presented to the Board for discussion. The MPB reviews each case to determine which specialized assays may yield meaningful insights to guide diagnosis, treatment, or monitoring. When warranted, the Board may cover part or all of the cost of the recommended test(s), helping to reduce barriers to equitable access.
The MPB does not replace the role of clinical care teams or existing tumour boards. Instead, it works in partnership with them to identify suitable patient cases and complement ongoing care with additional molecular testing opportunities that may not be available at the treating institution.
Ultimately, the National MPB supports a unified, national approach—helping ensure that patients, regardless of where they are treated, can benefit from the latest molecular diagnostics and expert interpretation to improve outcomes and advance research in childhood and young adult cancers.
Key Activities:
The MPB works to democratize access to advanced molecular assays by:
- Reviewing each referred case to identify the most appropriate assay(s)
- Improving visibility of available assays and creating clear pathways for clinicians to access them
- Bringing together a national expert panel that includes research, clinical, and lived-experience perspectives
- Providing financial support to cover the cost of Board-approved testing
- Building a pan-Canadian data registry to track demand for assays and understand access barriers
- Strengthening connections with pediatric and AYA cancer molecular tumour boards, test providers, and health-care partners across Canada
Submit a Patient Case to the MPB (For health care providers)
Clinician Submission Guide
Submit a Patient Case (Data Collection – Intake Form)
MPB-Approved List of Assays
Identify Assays to Add to the MPB-Approved List
Notify the MPB of an Advanced Molecular Assay
For more information, please contact Emily Nakada.
Principal Investigator: Philipp Lange
Chair: Cynthia Hawkins
Vice-Chair: Jonathan Bush
